Chemistry p element8/16/2023 The noble gases were characterized relatively late compared to other element groups. Periodic Trends All Group 15 elements tend to follow the general periodic trends: Electronegativity (the atoms ability of attracting electrons) decreases down the group. In the elemental form of the p-block elements, the highest energy electron occupies a p-orbital. The noble gases (Group 18) are located in the far right of the periodic table and were previously referred to as the 'inert gases' due to the fact that their filled valence shells (octets) make them extremely nonreactive. The nitrogen family is located in the p-block in Group 15, as shown below. Because its 2n shell is filled, it is energetically stable as a single atom and will rarely form chemical bonds with other atoms. P-BLOCK The elements consists of the last six groups minus helium (which is located in the s-block). For instance, lithium ( Li \text Ne start text, N, e, end text ), on the other hand, has a total of ten electrons: two are in its innermost 1 s 1s 1 s 1, s orbital and eight fill the second shell-two each in the 2 s 2s 2 s 2, s and three p p p p orbitals, 1 s 2 1s^ 2 1 s 2 1, s, squared 2 s 2 2s^ 2 2 s 2 2, s, squared 2 p 6 2p^6 2 p 6 2, p, start superscript, 6, end superscript. Elements in the second row of the periodic table place their electrons in the 2n shell as well as the 1n shell. After the 1 s 1s 1 s 1, s orbital is filled, the second electron shell begins to fill, with electrons going first into the 2 s 2s 2 s 2, s orbital and then into the three p p p p orbitals. This student friendly book has 15 chapters in which chemistry of the p-block elements is discussed in detail. The second electron shell, 2n, contains another spherical s s s s orbital plus three dumbbell-shaped p p p p orbitals, each of which can hold two electrons. Each element of the p-block is unique with respect to its properties and applications and their chemistry has undergone tremendous progress recently which this book brings out emphatically and with clarity. Hydrogen and helium are the only two elements that have electrons exclusively in the 1 s 1s 1 s 1, s orbital in their neutral, non-charged, state. On the periodic table, hydrogen and helium are the only two elements in the first row, or period, which reflects that they only have electrons in their first shell. This is written out as 1 s 2 1s^ 2 1 s 2 1, s, squared, referring to the two electrons of helium in the 1 s 1s 1 s 1, s orbital. Helium has two electrons, so it can completely fill the 1 s 1s 1 s 1, s orbital with its two electrons. This can be written out in a shorthand form called an electron configuration as 1 s 1 1s^ 1 1 s 1 1, s, start superscript, 1, end superscript, where the superscripted 1 refers to the one electron in the 1 s 1s 1 s 1, s orbital. Hydrogen has just one electron, so it has a single spot in the 1 s 1s 1 s 1, s orbital occupied. 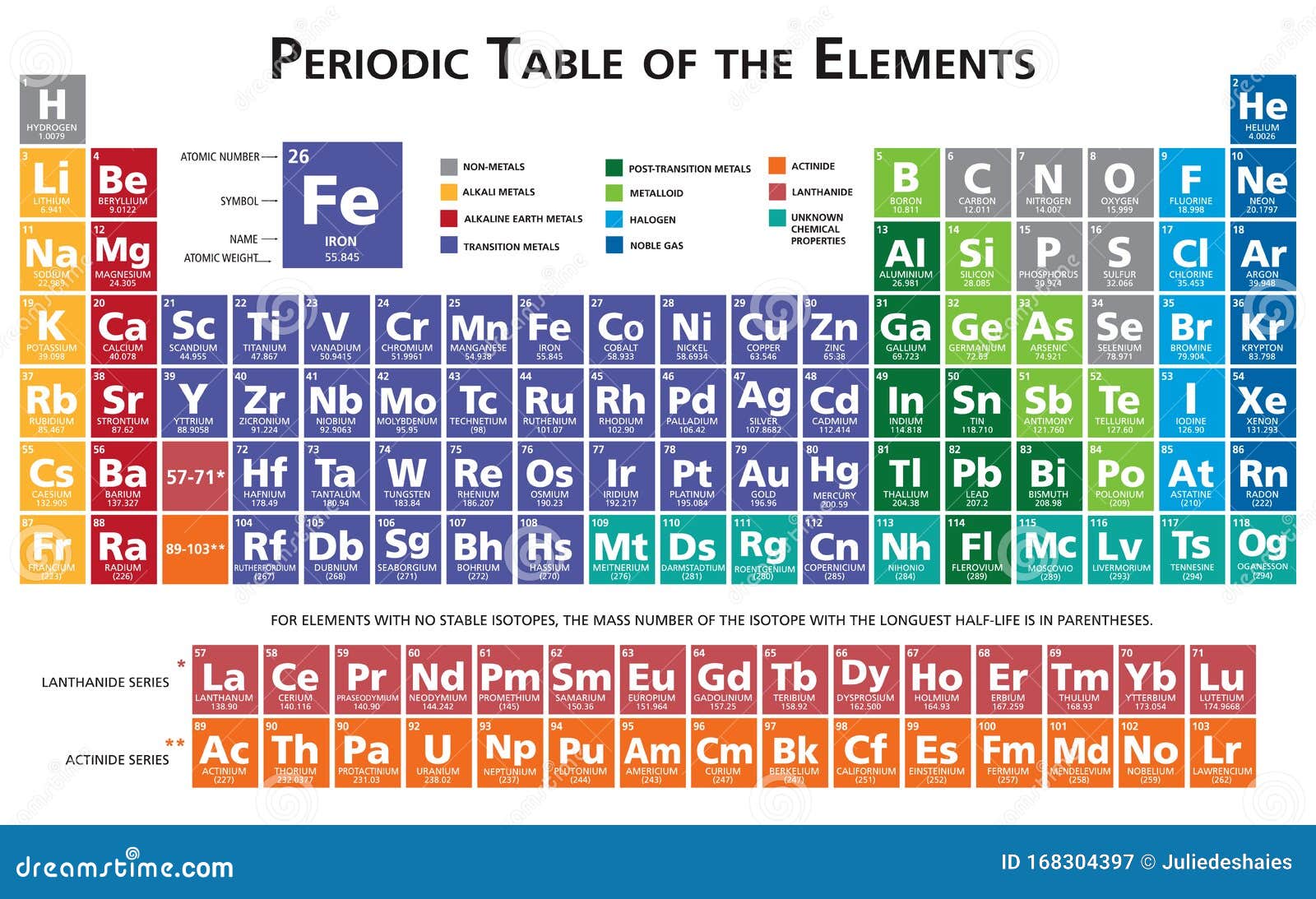
The 1 s 1s 1 s 1, s orbital is the closest orbital to the nucleus, and it fills with electrons first, before any other orbital. The first electron shell, 1n, corresponds to a single 1 s 1s 1 s 1, s orbital.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
